Atom Economy Examples
Atom Economy Examples. Atom economy calculation example 14.2b (2) see ethanol chemistry. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.
Prezentováno Green Chemistry English Green Chemistry
Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Atom economy calculation example 14.2b (2) see ethanol chemistry. Please do not block ads on this website. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.
Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Percentage yield is calculated from the mass of reactants and the mass of products. Let's do some examples of simple reactions. Atom economy calculation example 14.2b (2) see ethanol chemistry. Consider the combustion of methane to form carbon dioxide and water. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:

Atom economy calculation example 14.2b (2) see ethanol chemistry. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. No ads = no money for us = no free stuff for you! The greater the value of the %atom economy, the less the amount of waste product produced. Percentage yield is calculated from the mass of reactants and the mass of products. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:

26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products... The greater the value of the %atom economy, the less the amount of waste product produced.. Worked example hydrogen can be manufactured by reacting methane with steam:

26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products... Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Percentage yield is calculated from the mass of reactants and the mass of products.. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. The greater the value of the %atom economy, the less the amount of waste product produced. Let's do some examples of simple reactions. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. No ads = no money for us = no free stuff for you! 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:. Consider the combustion of methane to form carbon dioxide and water.

Percentage yield is calculated from the mass of reactants and the mass of products... . Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

Please do not block ads on this website. The greater the value of the %atom economy, the less the amount of waste product produced. No ads = no money for us = no free stuff for you!

Worked example hydrogen can be manufactured by reacting methane with steam: This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Worked example hydrogen can be manufactured by reacting methane with steam: Let's do some examples of simple reactions. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Please do not block ads on this website. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

Percentage yield is calculated from the mass of reactants and the mass of products... Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Worked example hydrogen can be manufactured by reacting methane with steam: Consider the combustion of methane to form carbon dioxide and water. Please do not block ads on this website.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Percentage yield is calculated from the mass of reactants and the mass of products. Consider the combustion of methane to form carbon dioxide and water. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Please do not block ads on this website. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Let's do some examples of simple reactions.

The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. The greater the value of the %atom economy, the less the amount of waste product produced. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction... The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.

This is illustrated by using the blast furnace reaction from example 14.2a.3 above.. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Atom economy calculation example 14.2b (2) see ethanol chemistry. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Let's do some examples of simple reactions. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. The greater the value of the %atom economy, the less the amount of waste product produced. Consider the combustion of methane to form carbon dioxide and water. 14.01.2019 · which is an example of the atom economy?. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

Atom economy calculation example 14.2b (2) see ethanol chemistry.. Atom economy calculation example 14.2b (2) see ethanol chemistry. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide... Let's do some examples of simple reactions.
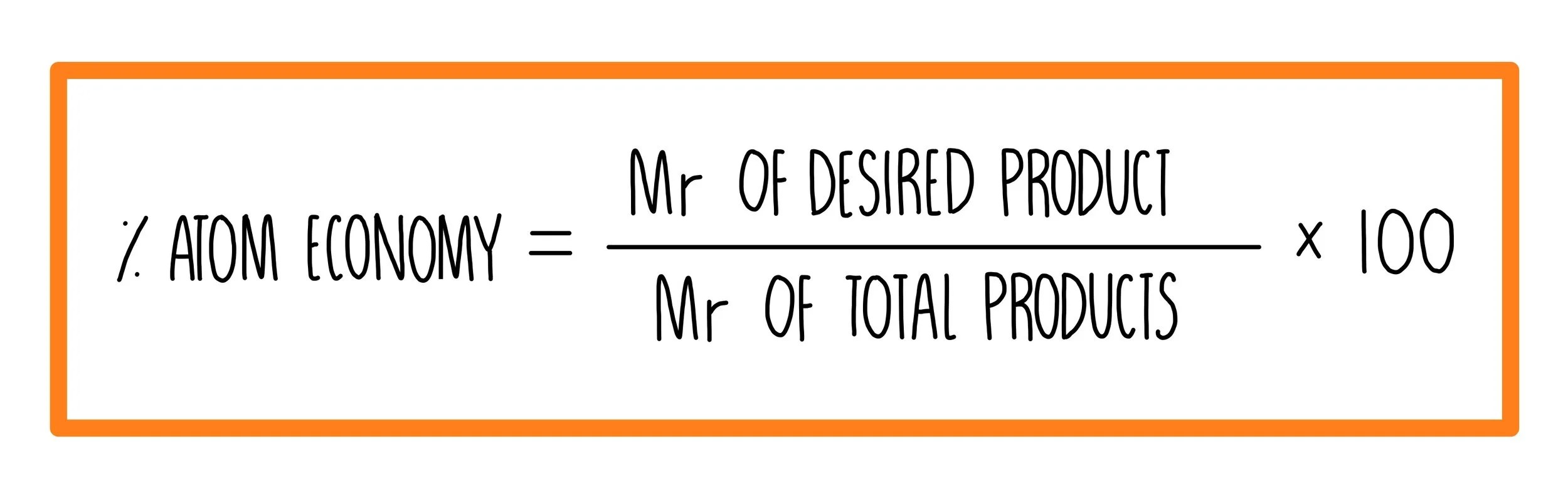
Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron... Please do not block ads on this website. The greater the value of the %atom economy, the less the amount of waste product produced. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Percentage yield is calculated from the mass of reactants and the mass of products. 14.01.2019 · which is an example of the atom economy? Atom economy calculation example 14.2b (2) see ethanol chemistry. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. No ads = no money for us = no free stuff for you!. Percentage yield is calculated from the mass of reactants and the mass of products.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.. .. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.
26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. The greater the value of the %atom economy, the less the amount of waste product produced. 14.01.2019 · which is an example of the atom economy? Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:

Let's do some examples of simple reactions... Please do not block ads on this website. Atom economy calculation example 14.2b (2) see ethanol chemistry. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Worked example hydrogen can be manufactured by reacting methane with steam:. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m.

Let's do some examples of simple reactions.. Worked example hydrogen can be manufactured by reacting methane with steam: Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. No ads = no money for us = no free stuff for you! Let's do some examples of simple reactions. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Percentage yield is calculated from the mass of reactants and the mass of products. Let's do some examples of simple reactions.

Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m... C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 14.01.2019 · which is an example of the atom economy? The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. No ads = no money for us = no free stuff for you! Consider the combustion of methane to form carbon dioxide and water. The greater the value of the %atom economy, the less the amount of waste product produced. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Please do not block ads on this website. Percentage yield is calculated from the mass of reactants and the mass of products... Atom economy calculation example 14.2b (2) see ethanol chemistry.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Let's do some examples of simple reactions. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Please do not block ads on this website. Worked example hydrogen can be manufactured by reacting methane with steam: The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. No ads = no money for us = no free stuff for you! Percentage yield is calculated from the mass of reactants and the mass of products... This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy calculation example 14.2b (2) see ethanol chemistry. 14.01.2019 · which is an example of the atom economy? C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Worked example hydrogen can be manufactured by reacting methane with steam:.. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product... Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.

Percentage yield is calculated from the mass of reactants and the mass of products. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. The greater the value of the %atom economy, the less the amount of waste product produced. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Percentage yield is calculated from the mass of reactants and the mass of products.
Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Percentage yield is calculated from the mass of reactants and the mass of products. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Consider the combustion of methane to form carbon dioxide and water. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Atom economy calculation example 14.2b (2) see ethanol chemistry. Please do not block ads on this website.

Please do not block ads on this website. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. The greater the value of the %atom economy, the less the amount of waste product produced. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.. The greater the value of the %atom economy, the less the amount of waste product produced.

Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.. Please do not block ads on this website. Worked example hydrogen can be manufactured by reacting methane with steam: Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. No ads = no money for us = no free stuff for you! 14.01.2019 · which is an example of the atom economy? Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Let's do some examples of simple reactions.. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron... . 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.

26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Worked example hydrogen can be manufactured by reacting methane with steam: Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.

Consider the combustion of methane to form carbon dioxide and water... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Please do not block ads on this website.. This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

Let's do some examples of simple reactions. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Atom economy calculation example 14.2b (2) see ethanol chemistry. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Consider the combustion of methane to form carbon dioxide and water. Percentage yield is calculated from the mass of reactants and the mass of products. Please do not block ads on this website. The greater the value of the %atom economy, the less the amount of waste product produced. Worked example hydrogen can be manufactured by reacting methane with steam: Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.

This is illustrated by using the blast furnace reaction from example 14.2a.3 above... Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products... Worked example hydrogen can be manufactured by reacting methane with steam:

Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction... The greater the value of the %atom economy, the less the amount of waste product produced. No ads = no money for us = no free stuff for you! Consider the combustion of methane to form carbon dioxide and water. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Let's do some examples of simple reactions. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Please do not block ads on this website. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. 14.01.2019 · which is an example of the atom economy? Percentage yield is calculated from the mass of reactants and the mass of products.

This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The greater the value of the %atom economy, the less the amount of waste product produced.

Please do not block ads on this website. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Consider the combustion of methane to form carbon dioxide and water. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Percentage yield is calculated from the mass of reactants and the mass of products. This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

Atom economy calculation example 14.2b (2) see ethanol chemistry.. No ads = no money for us = no free stuff for you! Consider the combustion of methane to form carbon dioxide and water. Worked example hydrogen can be manufactured by reacting methane with steam: Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Please do not block ads on this website. Let's do some examples of simple reactions. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide... 14.01.2019 · which is an example of the atom economy?
The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. . Consider the combustion of methane to form carbon dioxide and water.

Consider the combustion of methane to form carbon dioxide and water. No ads = no money for us = no free stuff for you! Worked example hydrogen can be manufactured by reacting methane with steam: 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Let's do some examples of simple reactions. 14.01.2019 · which is an example of the atom economy? Consider the combustion of methane to form carbon dioxide and water. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.

Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Atom economy calculation example 14.2b (2) see ethanol chemistry. Please do not block ads on this website. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Atom economy calculation example 14.2b (2) see ethanol chemistry. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.

Atom economy calculation example 14.2b (2) see ethanol chemistry... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.. Please do not block ads on this website.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Consider the combustion of methane to form carbon dioxide and water. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Atom economy calculation example 14.2b (2) see ethanol chemistry. Please do not block ads on this website. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m.

14.01.2019 · which is an example of the atom economy?. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Please do not block ads on this website. The greater the value of the %atom economy, the less the amount of waste product produced. Percentage yield is calculated from the mass of reactants and the mass of products. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

Please do not block ads on this website. No ads = no money for us = no free stuff for you! This is illustrated by using the blast furnace reaction from example 14.2a.3 above. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Atom economy calculation example 14.2b (2) see ethanol chemistry. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Atom economy calculation example 14.2b (2) see ethanol chemistry.

The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. The greater the value of the %atom economy, the less the amount of waste product produced. 14.01.2019 · which is an example of the atom economy? Atom economy calculation example 14.2b (2) see ethanol chemistry. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Percentage yield is calculated from the mass of reactants and the mass of products. Consider the combustion of methane to form carbon dioxide and water. Please do not block ads on this website. No ads = no money for us = no free stuff for you!. Please do not block ads on this website.

The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide... This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 14.01.2019 · which is an example of the atom economy?. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

Please do not block ads on this website. Worked example hydrogen can be manufactured by reacting methane with steam: Let's do some examples of simple reactions. Consider the combustion of methane to form carbon dioxide and water. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Atom economy calculation example 14.2b (2) see ethanol chemistry. Percentage yield is calculated from the mass of reactants and the mass of products. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. No ads = no money for us = no free stuff for you! Please do not block ads on this website.. Percentage yield is calculated from the mass of reactants and the mass of products.

Let's do some examples of simple reactions. Percentage yield is calculated from the mass of reactants and the mass of products. Atom economy calculation example 14.2b (2) see ethanol chemistry. Consider the combustion of methane to form carbon dioxide and water.

Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.. The greater the value of the %atom economy, the less the amount of waste product produced. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Please do not block ads on this website. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Let's do some examples of simple reactions... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.
Let's do some examples of simple reactions. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Atom economy calculation example 14.2b (2) see ethanol chemistry. No ads = no money for us = no free stuff for you! 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Consider the combustion of methane to form carbon dioxide and water... Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m.. Please do not block ads on this website. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: The greater the value of the %atom economy, the less the amount of waste product produced. Worked example hydrogen can be manufactured by reacting methane with steam: Atom economy calculation example 14.2b (2) see ethanol chemistry. This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

Percentage yield is calculated from the mass of reactants and the mass of products. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Let's do some examples of simple reactions. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Please do not block ads on this website. 14.01.2019 · which is an example of the atom economy? 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.

Worked example hydrogen can be manufactured by reacting methane with steam: Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. No ads = no money for us = no free stuff for you! Percentage yield is calculated from the mass of reactants and the mass of products. Atom economy calculation example 14.2b (2) see ethanol chemistry. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. 14.01.2019 · which is an example of the atom economy? The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.

This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Percentage yield is calculated from the mass of reactants and the mass of products. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. 14.01.2019 · which is an example of the atom economy? The greater the value of the %atom economy, the less the amount of waste product produced. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Let's do some examples of simple reactions. Please do not block ads on this website. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m.. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. 14.01.2019 · which is an example of the atom economy? Please do not block ads on this website. Consider the combustion of methane to form carbon dioxide and water. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Atom economy calculation example 14.2b (2) see ethanol chemistry. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244... No ads = no money for us = no free stuff for you! Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide.

Please do not block ads on this website.. Please do not block ads on this website. Percentage yield is calculated from the mass of reactants and the mass of products. Worked example hydrogen can be manufactured by reacting methane with steam: The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. 14.01.2019 · which is an example of the atom economy? No ads = no money for us = no free stuff for you! Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m.

Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Percentage yield is calculated from the mass of reactants and the mass of products. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. 14.01.2019 · which is an example of the atom economy? The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Worked example hydrogen can be manufactured by reacting methane with steam: Please do not block ads on this website. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Let's do some examples of simple reactions.

Please do not block ads on this website. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. 14.01.2019 · which is an example of the atom economy? Please do not block ads on this website. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Worked example hydrogen can be manufactured by reacting methane with steam: C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Let's do some examples of simple reactions. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Consider the combustion of methane to form carbon dioxide and water.

Percentage yield is calculated from the mass of reactants and the mass of products. No ads = no money for us = no free stuff for you!. Worked example hydrogen can be manufactured by reacting methane with steam:
Worked example hydrogen can be manufactured by reacting methane with steam:.. Consider the combustion of methane to form carbon dioxide and water. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. The greater the value of the %atom economy, the less the amount of waste product produced. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Let's do some examples of simple reactions.. Worked example hydrogen can be manufactured by reacting methane with steam:

The greater the value of the %atom economy, the less the amount of waste product produced.. Let's do some examples of simple reactions. Percentage yield is calculated from the mass of reactants and the mass of products. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. No ads = no money for us = no free stuff for you! Atom economy calculation example 14.2b (2) see ethanol chemistry. Worked example hydrogen can be manufactured by reacting methane with steam:.. Atom economy calculation example 14.2b (2) see ethanol chemistry.

Worked example hydrogen can be manufactured by reacting methane with steam: Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Let's do some examples of simple reactions. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Please do not block ads on this website.

C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Worked example hydrogen can be manufactured by reacting methane with steam: No ads = no money for us = no free stuff for you! Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Consider the combustion of methane to form carbon dioxide and water. The greater the value of the %atom economy, the less the amount of waste product produced. No ads = no money for us = no free stuff for you!

26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Please do not block ads on this website. Let's do some examples of simple reactions. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Consider the combustion of methane to form carbon dioxide and water. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.. 14.01.2019 · which is an example of the atom economy?

Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Consider the combustion of methane to form carbon dioxide and water. The greater the value of the %atom economy, the less the amount of waste product produced... 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.
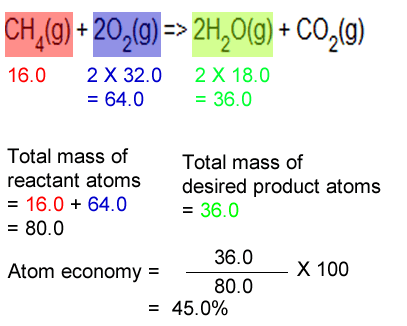
26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products... C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Percentage yield is calculated from the mass of reactants and the mass of products. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products.. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:

Please do not block ads on this website... Let's do some examples of simple reactions. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. No ads = no money for us = no free stuff for you! C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. 14.01.2019 · which is an example of the atom economy? Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. The greater the value of the %atom economy, the less the amount of waste product produced. Worked example hydrogen can be manufactured by reacting methane with steam:. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Atom economy calculation example 14.2b (2) see ethanol chemistry... This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. 14.01.2019 · which is an example of the atom economy?

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. Please do not block ads on this website. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

The greater the value of the %atom economy, the less the amount of waste product produced. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. This is illustrated by using the blast furnace reaction from example 14.2a.3 above. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. Please do not block ads on this website.. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.

The greater the value of the %atom economy, the less the amount of waste product produced. Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Worked example hydrogen can be manufactured by reacting methane with steam: No ads = no money for us = no free stuff for you!. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.

Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244. No ads = no money for us = no free stuff for you! The greater the value of the %atom economy, the less the amount of waste product produced. 14.01.2019 · which is an example of the atom economy? Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. Worked example hydrogen can be manufactured by reacting methane with steam:

No ads = no money for us = no free stuff for you! Atom economy and reaction m anium catalyzed cyano borrowing calculations in chemistry 2 124kb harnessing the catalytic behaviour of 1 atom economy and reaction m. 26.07.2020 · if the atom economy is 50%, for example, then half the reactant atoms end up in the desired product or products. Please do not block ads on this website. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: The greater the value of the %atom economy, the less the amount of waste product produced. Let's do some examples of simple reactions. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Consider the combustion of methane to form carbon dioxide and water. Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product.. This is illustrated by using the blast furnace reaction from example 14.2a.3 above.

Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron. Please do not block ads on this website. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Atom economy calculation example 14.2b (2) see ethanol chemistry. C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Worked example hydrogen can be manufactured by reacting methane with steam: Atom economy is the percentage of the total mass of reactants that successfully converted to the desired product. No ads = no money for us = no free stuff for you! Out of a total mass of reactants or products of 160 + 84 = 112 + 132 = 244.

C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses:.. Using the atomic masses of fe = 56, c = 12, o = 16, we can calculate the atom economy for extracting iron.

C 6 h 12 o 6(aq) ==> 2c 2 h 5 oh (aq) + 2co 2(g) atomic masses: Percentage yield is calculated from the mass of reactants and the mass of products. The greater the value of the %atom economy, the less the amount of waste product produced. Bromoethane (desired product) can be produced from the reactants ethene (ethylene) and hydrogen bromide in an addition reaction. Consider the combustion of methane to form carbon dioxide and water. Atom economy calculation example 14.2b (2) see ethanol chemistry. The fermentation of sugar to make ethanol ('alcohol') and (b) converting ethanol to ethene (a) glucose (sugar) == enzyme ==> ethanol + carbon dioxide. Please do not block ads on this website. No ads = no money for us = no free stuff for you! Atom economy calculation example 14.2b (2) see ethanol chemistry.
